Co-Chairs: Brad Elder, MD and Gavin Dunn, MD
Members: Dan Cahill, MD; Melanie Hayden Gephart, MD; Mark Linskey, MD; Timothy Smith, MD; Andrew Sloan, MD and Michael Vogelbaum, MD
The Clinical Trials and Registries sub-committee is excited to provide updates on important tumor registry and clinical trial activity. In addition to this update, we are planning to send a survey to neurosurgeons who specialize in neurosurgical oncology. This survey aims to gauge involvement and interest in national clinical trials and tumor outcomes registries. The survey should be brief, and we are targeting the maximal engagement of our peers. Please look for this survey in your email in the coming months.
Clinical Trials in Neuro-Oncology
In this report, we highlight an NRG study NRG-BN012, “A randomized phase III trial of pre-operative compared to post-operative stereotactic radiosurgery in patients with resectable brain metastases”. Inclusion criteria are patients with one to four brain metastases, which are candidates for SRS (12-20 Gy in a single fraction) and one of which requires surgical resection. The primary objective is PFS and time to AEs including radiation necrosis and LMD. Patients are randomized 1:1 to preop versus postop SRS. The target enrollment is 236 patients. PI is Stuart Burri, MD.
Further information regarding this clinical trial can be obtained from the NRG website at or clinicaltrials.gov.
NeuroPoint Alliance
Background:
The NeuroPoint Alliance (NPA) was established in 2008 to improve the quality of care for neurosurgical patients through the acquisition and analysis of clinical outcomes data. The goals of the NPA include:
- Establish risk-adjusted national benchmarks for both the safety and effectiveness of neurosurgical and spine procedures.
- Allow practice groups and hospitals to analyze their individual morbidity and clinical outcomes in real-time.
- Generate both quality and efficacy data to support claims made to public and private payers.
- Demonstrate the comparative effectiveness of neurosurgical procedures.
- Facilitate essential multi-center trials and other cooperative clinical studies.
- Data has supported production of >100 manuscripts.
Tumor QOD:
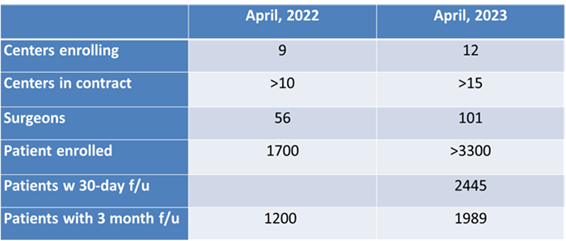
The NPA QOD Tumor Registry was developed in collaboration with the AANS/CNS Tumor Section Executive Committee and opened in April 2021 after a one-year pilot study. The Tumor Module focuses on outcomes for intracranial tumor procedures. Participating sites enter outcomes data online through the RedCap database system. Mayo Clinic is the coordinating center and provides data management, analysis and clinical and scientific guidance. The QOD Tumor Registry Steering Committee convened in 2021 provides strategic direction of the registry, provides content expertise, guidance and promotion of program activities. A quarterly report is provided to enrolling centers which gives a breakdown of demographics, case mix and outcomes in comparison to overall tumor registry data.
Participating in the Tumor QOD registry
Participating institutes can include academic medical centers, hospitals, ambulatory surgery centers, healthcare systems, private practice groups and individual surgeons. The startup process includes registration, training and onboarding, and is followed by continuous site support and monitoring.
The current price point is a three-year contract at $5,000/year registration fees, plus funding of 0.5 FTE for data entry. Patient accrual and startup of interested neurosurgical groups have increased steadily over the past year.
Those interested in more information regarding participating in the NPA Tumor QOD should contact:
Irene Zyung
Senior Manager, Registry Operations
Email: izyung@neuropoint.org
Phone: 847-378-0549